The probability of encountering instability increases when the size of the nucleus increases, since the mass of the nucleus becomes very large when it is concentrated. These forces are considered as extremely strong in nature. Two forces, namely the repulsive force, which is electrostatic, and the nucleus’s strong attractive forces hold the nucleus together. 
Energy is lost through radiation emitted by the unstable nucleus of an atom. Radioactivityĭue to the nuclear instability, the nucleus of an atom exhibits the phenomenon of radioactivity. 
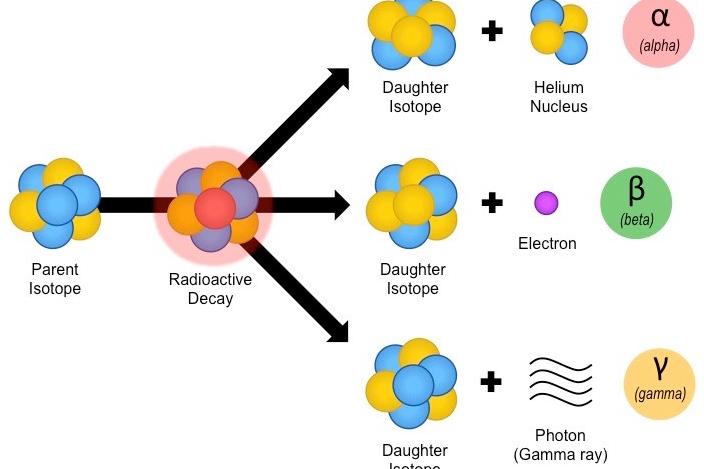
The mean-life is the sum of the lifespan of all atoms to the total number of atoms originally present. The lifespan of radioactive atoms therefore ranges from 0 to infinity. Thus, it is possible to determine the mean-life of a radioactive element. It obeys the exponential law and the lifetime of an atom can range from zero to infinity (∞). We know radioactivity is a spontaneous phenomenon. Though the intrinsic radioactivity is the same, the radiation dose received by someone handling a kilogram of high grade uranium ore will be much greater than for the same exposure to a kilogram of separated uranium, since the ore contains a number of short-lived decay products (see section on Radioactive Decay).Half – life or formerly known as Half – life period is one of the most common terminologies which is used in physics to define the radioactive decay of a specific sample or element over a specific period of time. The air in many 100 sq metre European homes (radon)ġ household smoke detector (with americium)ġ kg 50-year old vitrified high-level nuclear waste The air in a 100 sq metre Australian home (radon) Radioactivity of some natural and other materials Comparison * A former unit of (radio)activity is the Curie - 1 Bq is 27 x 10-12 curies. A becquerel is one atomic decay per second *. Gamma rays are often emitted with alpha or beta radiation also, as the nucleus decays to a less excited state.Īpart from the normal measures of mass and volume, the amount of radioactive material is given in becquerel (Bq), a measure which enables us to compare the typical radioactivity of some natural and other materials. The excited form is signified with an "m" (meta) beside its atomic number, eg technetium-99m (Tc-99m) decays to Tc-99. All the time, the atom is progressing in one or more steps towards a stable state where it is no longer radioactive.Īnother source of nuclear radioactivity is when one form of a radioisotope changes into another form, or isomer, releasing a gamma ray in the process. One can describe the emissions as gamma, beta and alpha radiation. If it decays with emission of an alpha or beta particle, it becomes a new element. When an atom of a radioisotope decays, it gives off some of its excess energy as radiation in the form of gamma rays or fast-moving sub-atomic particles. Some elements, eg uranium, have no stable isotopes. Unstable isotopes (which are thus radioactive) are called radioisotopes. This is called 'radioactive decay'.Įach element exists in the form of atoms with several different sized nuclei, called isotopes. An unstable atom has excess internal energy, with the result that the nucleus can undergo a spontaneous change towards a more stable form. These atoms are said to be 'unstable' or 'radioactive'. Most atoms are stable a carbon-12 atom for example remains a carbon-12 atom forever, and an oxygen-16 atom remains an oxygen-16 atom forever, but certain atoms eventually disintegrate into a totally new atom. Radiation comes from atoms, the basic building blocks of matter. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
